BIOL2019FINCH58955 BIOL
Investigating sex-based differences in pathogen resistance and immune responses in the fathead minnow (Pimephales promelas)
Type: Undergraduate
Author(s):
Miranda Finch
Biology
Lynsey Malin
Biology
Leah Thornton Hampton
Biology
Advisor(s):
Marlo Jeffries
Biology
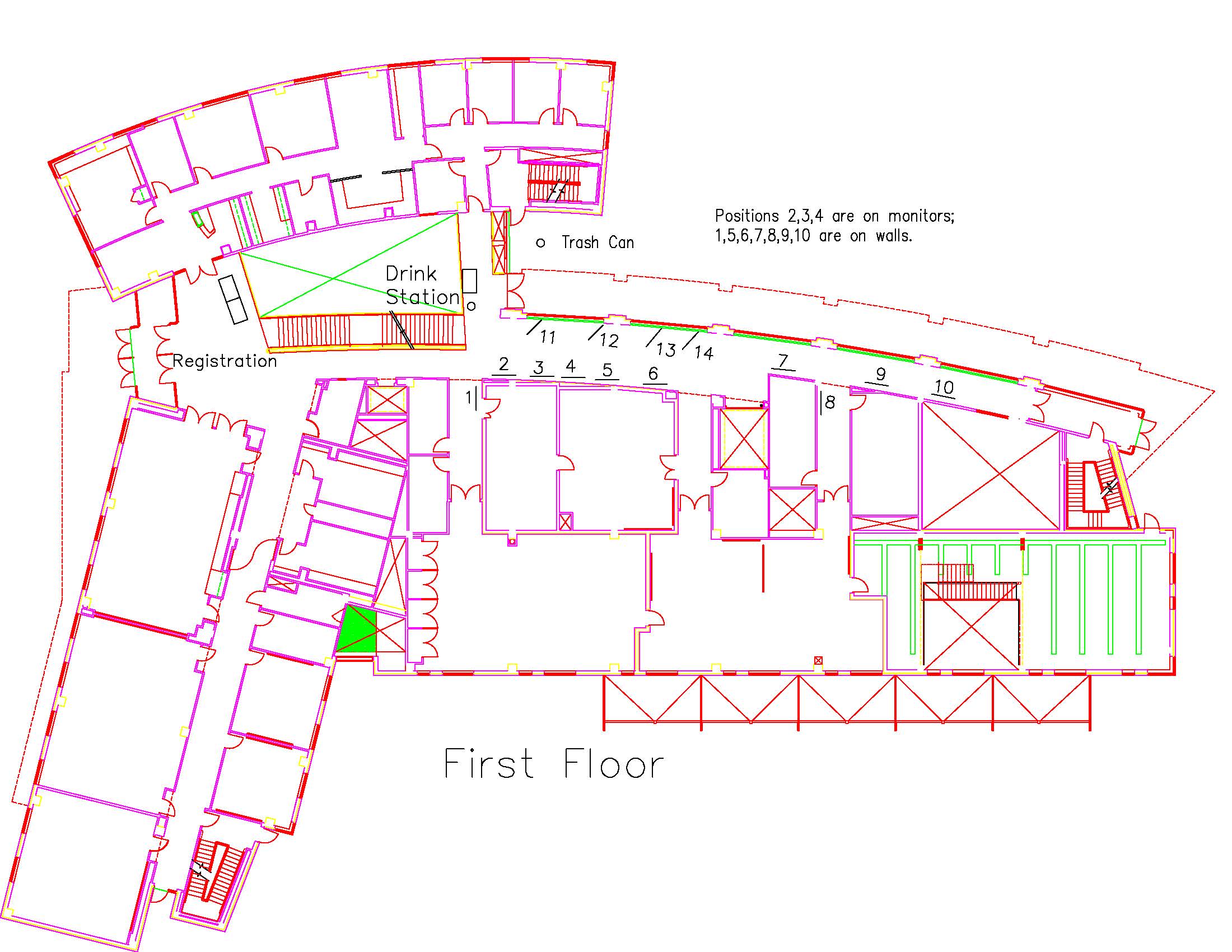
Location: Session: 1; 1st Floor; Table Number: 3
(Presentation is private)Males and females differ with regard to their immune response to a pathogen. Previous studies have observed males having reduced pathogen resistance. This suggests that they may be responding to pathogens differently. However, few studies have compared male and female immune responses following pathogen exposure. The purpose of this study was to examine sex-based differences in pathogen resistance and immune responses following exposure to a pathogen in adult fathead minnows (Pimephales promelas). To accomplish this, fish were bacterially infected with Yersinia ruckeri and the immune system’s ability to respond was monitored. Additionally, genes that are known to turn on during the immune response initiation were measured quantitatively providing insight into the molecular effect in minnows. At the whole organism level, male fish were less able to survive pathogen infection relative to female fish. At the tissue level, both male and female pathogen injected fish had decreased hematocrit percentages compared to the fish injected with a saline solution but did not differ from each other. At the molecular level, increased gene expression of interleukin 1β was seen in pathogen-injected males compared to pathogen-injected females and both sham-injected sexes indicating that pathogen-injected males mounted a larger inflammatory response at the molecular level. Taken together, this evidence suggests that the increased mortality observed among males earlier in the exposure to the pathogen may be due to the upregulated inflammatory response rather than the effects of the pathogen itself.
BIOL2019JORDAN10583 BIOL
Investigating the Role of Glymphatic Clearance of Amyloid Beta Through Exercise in C57BL/6J Mice
Type: Undergraduate
Author(s):
Rachel Jordan
Biology
Chris Hagen
Biology
Sofia Lopez
Biology
Julia Peterman
Psychology
Jordan White
Psychology
Advisor(s):
Michael Chumley
Biology
Gary Boehm
Psychology
Location: Session: 1; 1st Floor; Table Number: 7

View PresentationAlzheimer’s disease (AD) is a very prevalent neurodegenerative disorder and is the 6th leading cause of death in the United States. Alzheimer’s disease (AD) is characterized by widely distributed amyloid plaques and neurofibrillary tangles, and is clinically associated with a progressive decline in memory and other cognitive functions. The major protein component of neuritic plaques is amyloid beta peptide. Several pieces of evidence have indicated that amyloid beta accumulates to form oligomeric states in the AD brain and cause the cognitive dysfunction commonly seen in patients. While a decrease in cognitive function is considered a hallmark of the disease, AD patients also exhibit decreased motor abilities and difficulties learning new motor tasks. Our lab’s previous investigations found voluntary running to decreased amyloid beta burden in C57/BL6 mice. The present experiment seeks to further explore the mechanism through which exercise induced amyloid beta clearance occurs. Previous studies have pointed to the function of the glymphatic system in the clearance of amyloid beta. The level and distribution of aquaporin 4 (AQP4) in the vascular endfeet of astrocytes is crucial to the normal function of the glymphatic system. Our experiment sought to determine a more detailed analysis of the role of AQP4 in the glymphatic clearance of amyloid beta. Using TGN-020, a selective AQP4 antagonist, we hope to further determine the importance of glymphatic clearance of amyloid beta in C57/BL6 mice through exercise. We hypothesize that mice receiving intraperitoneal TGN injections, thus blocking the function of AQP4, will experience decreased glymphatic clearance of amyloid beta. Understanding the process of amyoid beta clearance can aid in treatments for both the pathological and clinical affects associated with AD.
BIOL2019LOSEFSKY29412 BIOL
The role of ClpX and ClpP in antibiotic resistance in Bacillus antracis
Type: Undergraduate
Author(s):
Quinn Losefsky
Biology
Advisor(s):
Shauna McGillivray
Biology
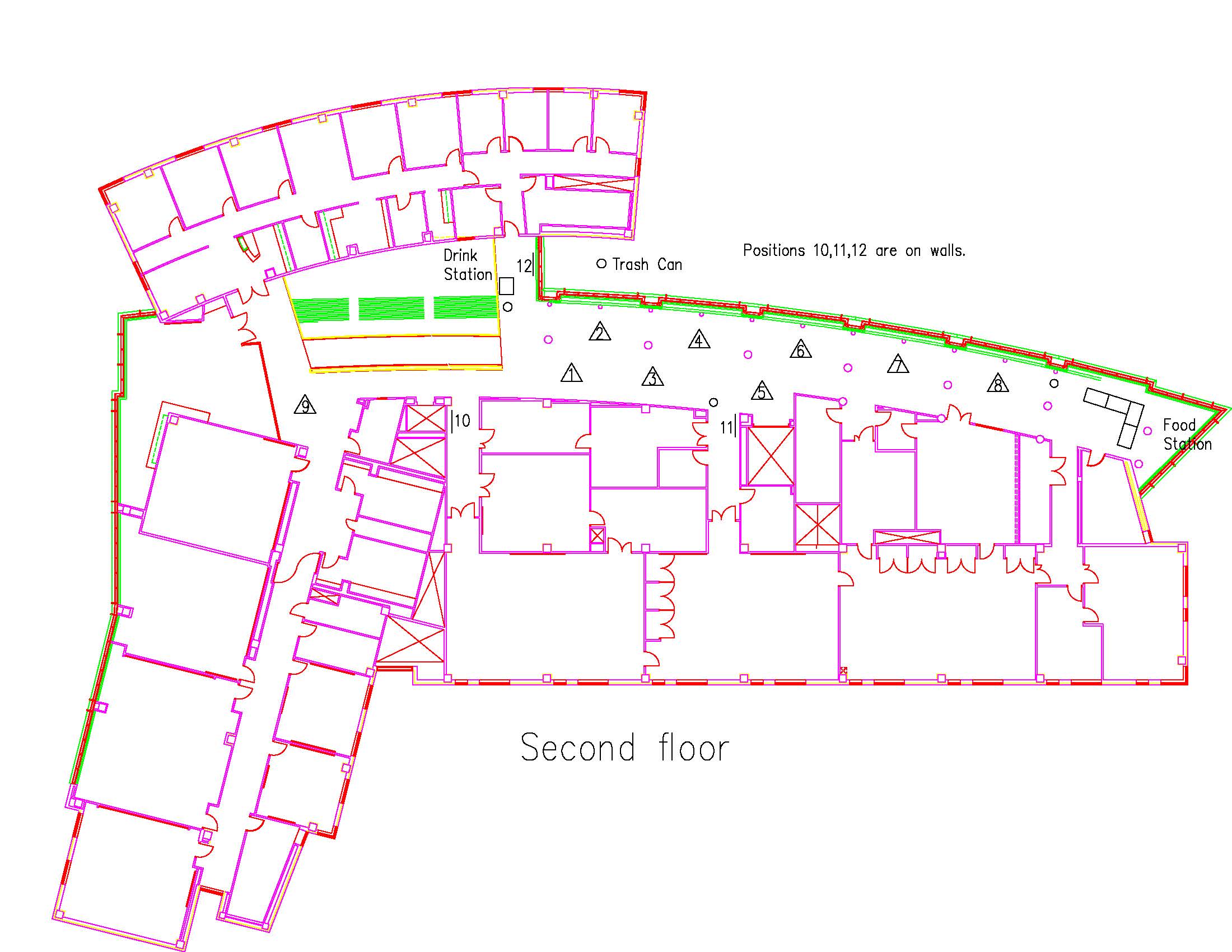
Location: Session: 1; 2nd Floor; Table Number: 4
View PresentationBacillus anthracis is a Gram-positive bacterium that causes anthrax in humans. It is a significant microorganism in that many proteins important to virulence or pathogenesis are highly conserved in many other pathogenic bacteria. Our lab has previously identified the protein ClpX in Bacillus anthracis as metabolically significant in antibiotic resistance. Specifically, B. anthracis lacking the ClpX gene (ΔClpX) are significantly more susceptible to antibiotics that target the bacterial cell wall such as penicillin than the wild type. ClpX has multiple functions; primarily it interacts with ClpP to form a proteolytic complex that degrades dysfunctional or obsolete proteins. ClpX also has an independent chaperone function, moving proteins around the cell. This project has focused on determining if the pathway of decreased antibiotic resistance in mutant B. anthracis is dependent on ClpX interactions with ClpP, or if ClpX can function independently. To test this, a point mutation (I265E) was made in the ClpX gene at the site that has been previously identified as the site of interaction between ClpX and ClpP in Staphylococcus aureus. The ClpX genes in B. anthracis and S. aureus exhibit a high degree of conservation particularly in this region, and it is expected that this site will also be critical for ClpX and ClpP interaction in B. anthracis. The mutated ClpX gene (I265E) has been confirmed with sequencing and has been transformed as an inducible expression plasmit into the ΔClpX B. anthracis strain. The next step is to perfom growth assays in antibiotics such as penicillin to ascertain if the mutated expression vector can restore antibiotic resistance to ΔClpX.
BIOL2019MOROTECOSTAS54682 BIOL
The Effect of BRCA1 Activation on Protein Structure and Function
Type: Undergraduate
Author(s):
Brian Morote-Costas
Chemistry & Biochemistry
Advisor(s):
Mikaela Stewart
Biology
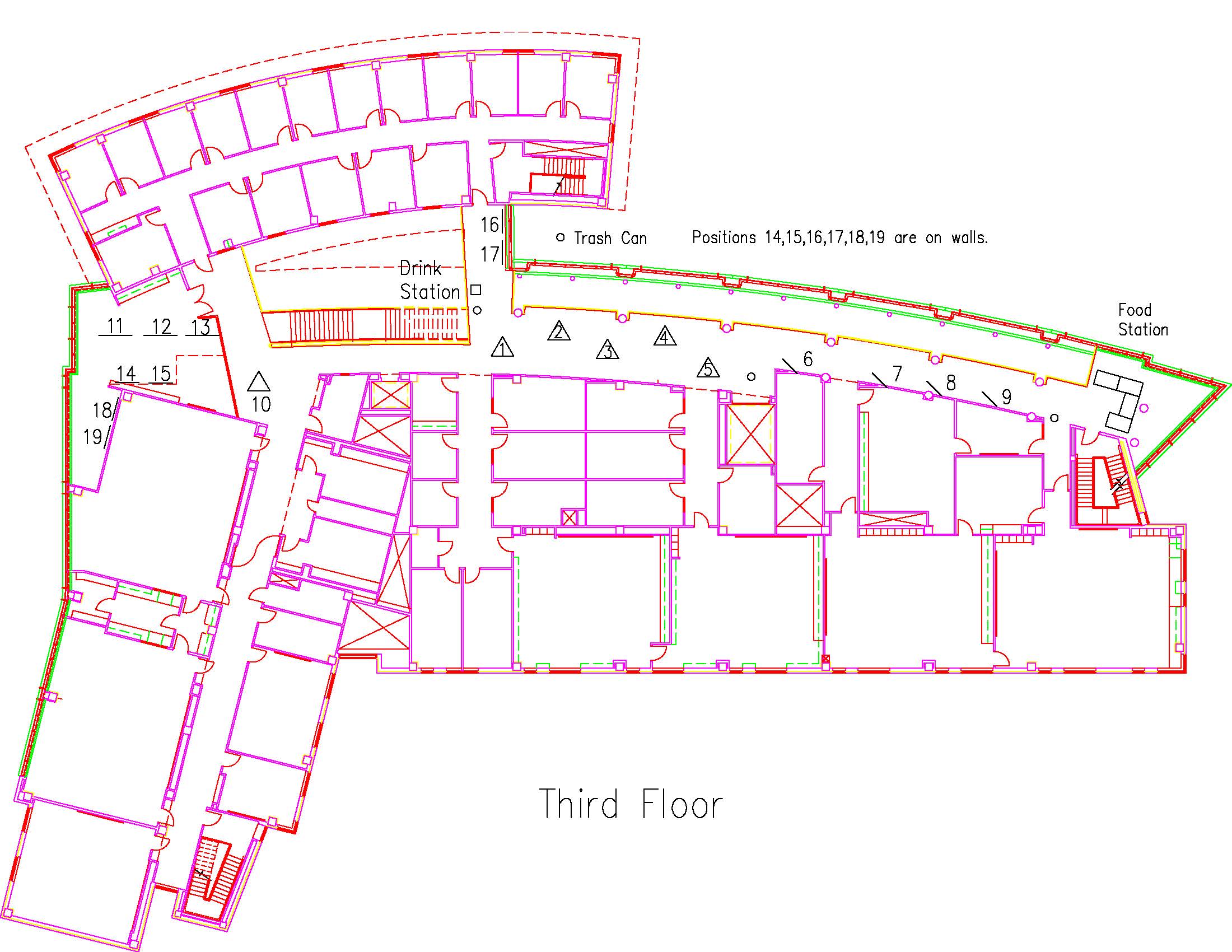
Location: Session: 1; 3rd Floor; Table Number: 3
View PresentationThe proper function of the Breast Cancer susceptibility gene 1 (BRCA1) and its subsequent protein in the human body is vital to healthy individuals. The malfunction of BRCA1 due to a mutation is associated with an increased risk of breast or ovarian cancer of up to 80%. The key to understanding whether a mutation in BRCA1 will lead to higher risk of cancer is its location. Many of its interactions with other proteins take place in the central region of BRCA1. Currently, little is known about how mutations located in this region affect BRCA1 structure and function. The central region is where BRCA1 interacts with another tumor suppressor called PALB2 to perform DNA repair. The central region site studied in this project can be activated as a response to DNA damage, influencing BRCA1 and PALB2 interaction to generate a damage repair response. We show how activation of BRCA1 affects its structure and function on the molecular level. The accomplish this we created three mutations in the central region that mimic activation of BRCA1 to identify possible significant changes in protein-protein interactions using biochemistry and structural biology techniques like Isothermal titration calorimetry and circular dichroism.
BIOL2019NAGEL36745 BIOL
Quantifying Synapses in a 5xFAD Mouse Model Using Immunolabeling and Tissue Clearing Techniques
Type: Undergraduate
Author(s):
Gabrielle Frediani
Biology
Kelly Brice
Psychology
Christopher Hagen
Biology
Briar Hill
Psychology
Sarah Nagel
Biology
Filza Qureshi
Psychology
Gabrielle Salinas
Psychology
Advisor(s):
Michael Chumley
Biology
Gary Boehm
Psychology
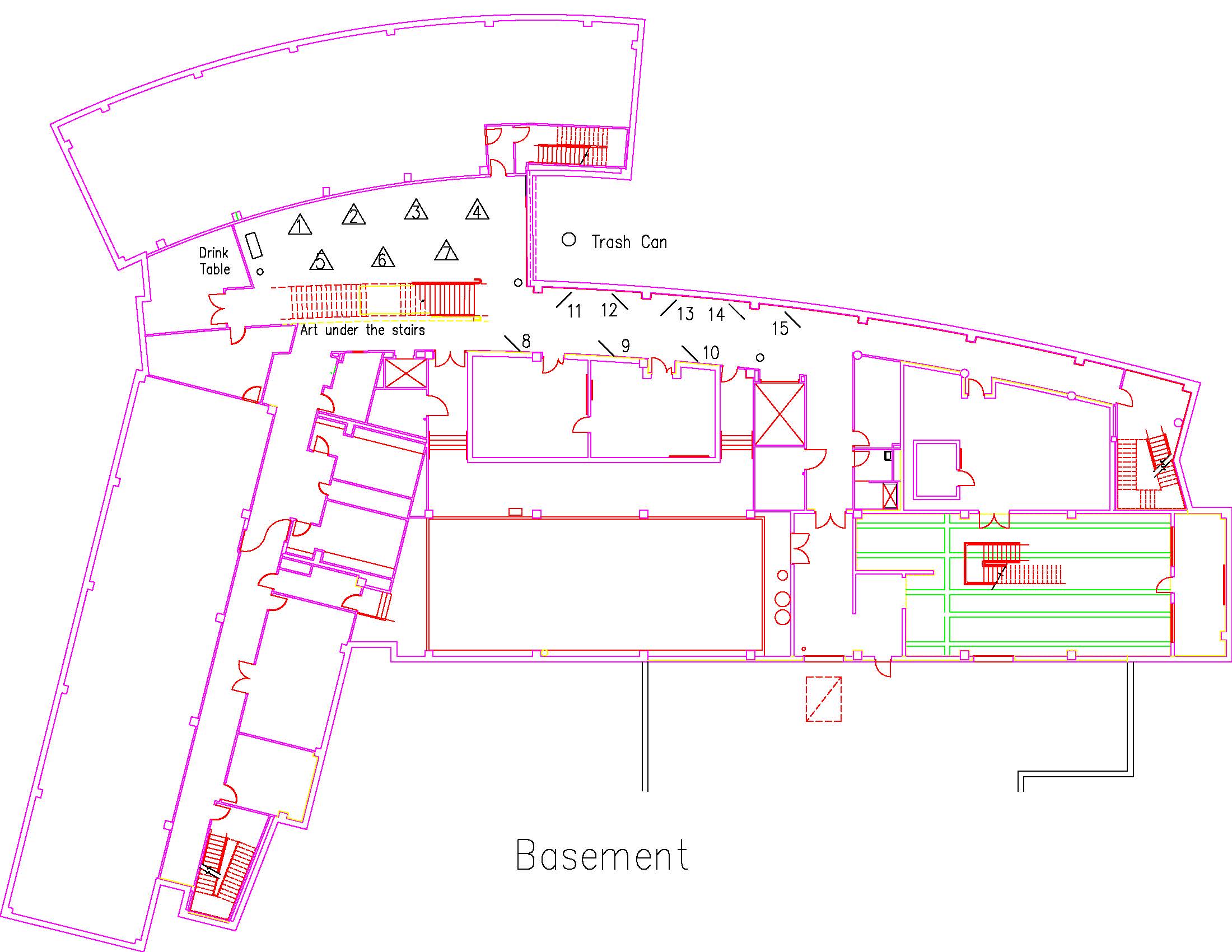
Location: Session: 2; Basement; Table Number: 4
View PresentationAlzheimer’s disease (AD) is a neurodegenerative disease resulting in dementia and memory impairment in patients. Research is immensely important as there are currently no treatments available to slow, prevent, or cure the damage inflicted upon individuals. The brain dysfunction in AD is marked by an increase in amyloid beta (Aβ), the protein responsible for plaque deposition in the brain. The severity of the cognitive deficits positively correlates with the load of Aβ. Prior research in animal models has pointed to Aβ causing synaptic disruption. A synapse is associated with information transfer inside the brain. Neuroscientists are trying to determine if the synapse disruption caused by Aβ leads to the cognitive dysfunction seen in AD. However, it is difficult it is to visualize individual neurons inside the hippocampus. In the present study, our aim was to understand the effect that Aβ has on synapses using an immunolabeling and tissue clearing technique in an AD transgenic mouse model. The 5xFAD mouse model utilized in this study rapidly develops Aβ pathology. This model mimics the pathophysiology of AD in humans. The mouse model used was also knock-in transgenic for Green Fluorescent Protein (GFP) on mature neurons. Using immunolabeling, GFP was tagged with antibodies, thus making neurons visible under the microscope. Antibodies were also used for Aβ in order to visualize the amount of Aβ protein and its location. Images of neurons were obtained in both FAD+/GFP+ and FAD-/GFP+ mice using a conventional staining method and a tissue clearing technique. We expected the tissue clearing technique, which clears lipids from the brain, to result in enhanced visualization of neurons but this was not the case. To quantify synapses, dendritic spines were counted on individual neurons in both groups of mice. Our results showed that Aβ accumulation in FAD+ mice correlates with a decreased number of dendritic spines signifying a decrease in synaptic density.